Intacs
Intacs may be used in keratoconus patients under certain circumstances.
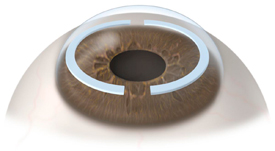
This procedure is FDA approved and involves placing circular segments within the peripheral cornea. The segments are at a depth and location where they can provide support and stability of the peripheral cornea while allowing the patient to see through the central portion of their own corneal.



Intacs may be recommended in cases of moderate to advanced keratoconus and may delay or eliminate the need of a corneal transplant.
![]()
